The Path and Method of Collagen Filler Entering the International Market
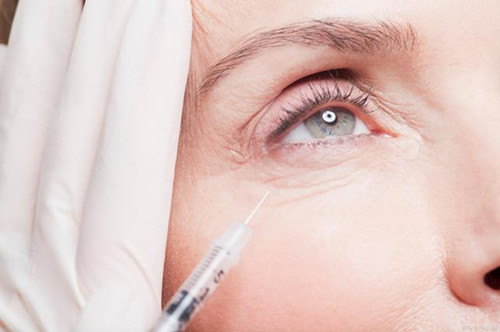
Collagen is a biopolymer and the main component of animal connective tissue. It is also the most abundant and widely distributed functional protein in mammals, accounting for 25% ~ 30% of the total protein. Skin is the largest organ of the human body. 72% of the skin is collagen, and 80% of the dermis. As a structural protein of extracellular matrix (ECM), collagen consists of three polypeptide chains to form a three strand helical structure. Many collagen macromolecules can form a fiber cross-linked structure side by side. It has high mechanical strength and acts as a "scaffold" in the dermis of the skin. With the growth of age, the loss of collagen accelerates and wrinkles gradually appear on the skin. Therefore, injecting collagen products is the most direct way to supplement the lost collagen and improve wrinkles.
It has been 40 years since the world's first collagen filler was launched in 1981. Its safety and effectiveness have been widely and long-term verified. With the continuous innovation and improvement of products and production processes, collagen fillers have ushered in great prospects. The products developed with collagen from different sources also show obvious differences in clinical application. Mammalian collagen is widely used because its source is easy to obtain, consistent with human living environment and easy to preserve, transport and use. Its protein structure has high homology with human body and has the biological activity of natural collagen. How does China's collagen filler enter the international market? Zhejiang Chumsun Biological Products Co., Ltd. continued to pay attention to international trends, sorted and summarized the laws and regulations of EU, the United States, ASEAN and other countries and regions, and provided a reference path for domestic collagen fillers to go abroad.
1. EU registration
a) Regulations
The current EU regulation on medical devices is regulation (EU) 2017 / 745, referred to as "MDR". It was promulgated in 2017 and officially implemented in May 2021, replacing MDD (93 / 42 / EEC) and aimdd (90 / 385 / EEC), and bringing beauty products for medical purposes into medical devices for management for the first time. Medical device products need to be registered before they can be legally sold in the market.
b) Classification of collagen fillers
For the first time, MDR has included beauty products for non-medical purposes into the scope of medical devices for management: products without expected medical purposes listed in Annex XVI, such as beauty pupil, facial filling or injection, tattoo, skin improvement and beauty products.
Like most countries in the world, EU medical devices are classified and managed according to the risk level. The higher the level, the greater the risk. From MDD to MDR, instruments are still divided into four categories: Class I, Class IIa, Class IIB and Class III. Annex VIII RULE8 classifies collagen fillers into Class III: implantable invasive devices with biological effects or can be fully or mostly absorbed. In this case, they should be classified as Class III.
c) EU registration path for class III products
Under the MDR regulations, when overseas enterprises register and sell medical device products in the EU, they must authorize the only EU agent. Therefore, Chinese enterprises register medical device products in the EU as manufacturers, and the other two necessary roles are EU authorized agents and announcement agencies.
The roles and responsibilities of the European authorized representative are:
1. Provide the registered address in the EU and display the address of the EU agent on the product package;
2. Keep the updated EU doc, technical documents and certificates permanently at the registered address for inspection by the competent authorities;
3. Maintain a continuous and effective EU representative role in the European Commission, EU competent authorities, EU Nb and EU users to represent manufacturers;
4. Fill in applications such as equipment registration and EU free sales certificate on behalf of the manufacturer;
5. Deal with any incident (alert report) reported by medical staff, patients and users, and immediately notify the manufacturer;
6. Maintain compliance news and updates;
7. Check the registration of importers, manufacturers and products themselves, as well as the registration of the EU, and check and update it;
8. Consult and continue to support the manufacturer. If the manufacturer violates its obligations under EU MDR / ivdr, the authorization shall be terminated.
The responsibilities of the notified body are to review the compliance with laws and regulations (MDR or ivdr), including the review of the manufacturer's quality system, review the relevant technical documents provided by the manufacturer to prove its product safety and performance statement according to the specific classification of products, and issue CE certification.
After Chinese enterprises obtain the CE certificate, the products can be sold freely within the EU after being filed in the EU by the only European authorized agent designated by them.
The path of registering class III products in the EU: the manufacturer selects the announcement agency with the corresponding product review and certification qualification, and the announcement agency decides to accept it. First, the system audit shall be conducted on the manufacturer, then the product registration data shall be reviewed, and then the certificate shall be issued. Registration data trials need to be completed in oecd-glp Laboratory (CMA or CNAs in China), and clinical trials can accept clinical data in China.
d) Notified body that can accept the application for filler registration
Under the MDR regulations, the number of notified bodies that have passed the certification has been reduced from 49 original MDD certification to 23 MDR certification. Of the 23 notified bodies, only 19 can accept the registration application of Passive Implants - passive soft tissue and other implant products. Because the competent EU authorities have not issued the guidelines for the certification of announcement bodies of annex XVI products, none of the above 19 announcement bodies has passed the certification of annex XVI, and no announcement body is qualified to accept the registration of collagen fillers. With the improvement of relevant EU regulations and rules, the path for collagen fillers to enter the EU market will be further clarified.
2、 Registered in the United States
a) Regulations
The U.S. Food and Drug Administration (FDA) is the competent authority responsible for the registration of medical devices in the United States. It only allows safe and effective products to enter the market, and protects public health by monitoring their continuous safety after the product is put on the market.
The United States is the first country in the world to legally define "medical device", and the legislative supervision of medical devices has a history of more than 100 years. In the current regulatory system of the United States, the regulation on the management of medical devices is part 800-1299 of volume 8 of the code of federal regulations (CFR), medical device act.
b) Classification of collagen fillers
China is the first country to implement classified management of medical devices. According to the risk level from low to high, it is classified into products exempted from pre market notification, products requiring pre market notification (510k) and products requiring pre market approval (PMA). The product number of collagen filler classified by FDA is LMH (implant, dermal, for aesthetic use), and the registration method is PMA.
c) FDA registration path of medical devices
As mentioned above, the FDA headquarters reviews the medical devices according to the risk level of the products by pre marketing notification (510k) or pre marketing approval (PMA).
Most low-risk products can be registered according to the 510k path. The path is to register first and then go on sale. When the products have a certain popularity or high sales volume, the U.S. competent department will conduct a system audit on the manufacturer.
High risk products are registered according to the PMA path. The path is that the competent department first reviews the system of the manufacturer, then reviews the registration data, and the products can be sold only after they are approved for listing.
The clinical trial materials in the U.S. registration data shall comply with the EPA-GLP statement (CMA or CNAs in China). The ethnic differences shall be investigated in the clinical trial, that is, only part of the clinical trial data in China can be adopted.
3. Registration in other countries
ASEAN countries recognize the registration certificate in China, that is, after the medical device products are successfully registered in China, they can be registered or filed in ASEAN countries as long as they submit simple materials, and then they can be listed and sold legally. On the premise of obtaining CE certification in Russia and Brazil, the products can be approved for registration and listing only after completing clinical trials in Russia and Brazil.
REFERENCE:
[1] REGULATION (EU) 2017/745
[2] REGULATION (EU) No 722/2012
[3] WHO Guidelines on Tissue Infectivity Distribution in Transmissible Spongiform Encephalopathies (2006)
[4] Federal Food, Drug, and Cosmetic Act of 1938
[5] Code of Federal Regulations
Latest News
Contact Us

Name: International Trade Center
Tel: +86 18969974572
E-mail: [email protected]
Add: #54 Building, No. 650 Hongfeng Rd, Donghu Street, Linping District, Hangzhou, Zhejiang Province, 311100, P.R.China
 CHINESE
CHINESE ENGLISH
ENGLISH




 Mail inquiry
Mail inquiry